|
SafeMedicationUse Newsletter
|
|
The Power of Words: Improving Labels and Packages for Medication Safety
Part 3 of a 3-Part Series
2024-06-11
-
This newsletter is focused on the importance of clear information on labels to support decisions and selection of products for use. The first newsletter described the power of an informed consumer. The second newsletter described the power of strong relationships with health care providers. This third newsletter also describes the power of reporting when information on medication labels and packages is confusing.
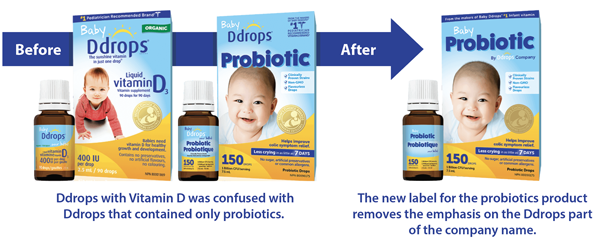
Mederror.ca received a report about a parent who intended to buy Baby Ddrops (vitamin D) for their child. However, the parent mistakenly bought a package labelled as Baby Ddrops Probiotic. Despite its name, the product purchased did not contain any vitamin D. ISMP Canada shared the incident with the company that makes the products and Health Canada. The company appreciated the reported concern and changed the label in less than 3 weeks to support safe use of their products.
The following tips can help make products and care safer:
-
If the information on the product package is unclear, talk to the pharmacist.
-
If you think information on a label or package might lead to a medication mistake, tell us about it at mederror.ca.
For more information on the important work that Health Canada is doing to increase the safety of health products visit: Natural health product regulation in Canada: Protecting Canadians from Unsafe Drugs Act (Vanessa's Law)
This newsletter was developed in collaboration with Best Medicines Coalition and Patients for Patient Safety Canada.
Recommendations are shared with healthcare providers, through the ISMP Canada Safety Bulletin, so that changes can be made together.
|

